Difference between revisions of "Novartis"
(SW: →External Resources: editttt) |
(SW: →Human Rights: edittttt) |
||
| Line 284: | Line 284: | ||
The report was released on Friday, 17 November during an event held to mark the silver jubilee of the Wemos Foundation. Liesbeth van der Kruit took such politicians as Diederik Samsom (PvdA) and Kees Vendrik(GroenLinks) to task about this issue during the Grote Verkiezingsshow (the Great Election Show). The report describes tests that involved various well-known pharmaceutical companies such as Novo Nordisk, Pfizer, Johnson & Johnson, Novartis, AstraZeneca and GlaxoSmithKline."<ref>[http://somo.nl/news-en/misdeeds-by-pharmaceutical-industry/?searchterm=novartis "Report from the Dutch Centre for Research on Multinational Corporations on unfair pharmaceutical trial practices"]</ref> | The report was released on Friday, 17 November during an event held to mark the silver jubilee of the Wemos Foundation. Liesbeth van der Kruit took such politicians as Diederik Samsom (PvdA) and Kees Vendrik(GroenLinks) to task about this issue during the Grote Verkiezingsshow (the Great Election Show). The report describes tests that involved various well-known pharmaceutical companies such as Novo Nordisk, Pfizer, Johnson & Johnson, Novartis, AstraZeneca and GlaxoSmithKline."<ref>[http://somo.nl/news-en/misdeeds-by-pharmaceutical-industry/?searchterm=novartis "Report from the Dutch Centre for Research on Multinational Corporations on unfair pharmaceutical trial practices"]</ref> | ||
| + | |||
| + | |||
| + | '''"Compulsory licensing “a last mechanism,” says Thai Health Minister"''' | ||
| + | |||
| + | '''August, 2008.''' "Thailand’s new Public Health Minister, Chawarat Charnveerakul, has said that imposing compulsory licensing (CL) is “the last mechanism” that the government would use “to help the poor get access to life-saving drugs at a fair price." | ||
| + | |||
| + | "First, we have to negotiate with drug firms to reduce the price of their drugs. If they do not agree with us we will then impose compulsory licensing for expensive drugs to save our patients’ lives," said Mr Chawarat, who was appointed to his post this month in a cabinet reshuffle. He added that his Ministry had not revoked the CL policy as a way of obtaining essential drugs, but retained it as “a key mechanism” in negotiating for lower prices."<ref>[http://pharmatimes.co.uk/Forums/forums/t/2065.aspx "Compulsory licensing on pharmaceutical drugs in Thailand"]</ref> | ||
===Environment === | ===Environment === | ||
Revision as of 05:32, 11 December 2008
|
This article is part of the Center for Media & Democracy's spotlight on global corporations. |
| Type | Private pharmaceutical company |
|---|---|
| Genre | Pharmaceutical drug producer and biotechnological researcher |
| Founded | 1996 |
| Headquarters | Basel, Switzerland |
| Employees | 67,653 |
Novartis is a pharmaceutical multinational from Switzerland that specializes in biotechnological drug production and distribution, as well as a vast array of consumer healthcare products, including the manufacturing of vaccines, the development of over-the-counter pharmaceuticals not requiring prescrptions, as well as animal healthcare products and the production of contact lenses and various lense care products. It is the world's 5th largest vaccine producer, and its presence is noted in over 140 countries worldwide through 100.000 or so associates[1]. The company formerly owned the Gerber Products Company, a major infant and baby products producer, but sold it to Nestlé on September, 2007.[2]
In addition to internal research and development activities, Novartis is also involved in publicly funded collaborative research projects, with other industrial and academic partners. One example in the area of non-clinical safety assessment is the InnoMed PredTox. The company is also expanding its activities in joint research projects within the framework of the Innovative Medicines Initiative of EFPIA and the European Commission.
Contents
Company History
"Novartis was founded in 1996 with the merger of Ciba-Geigy Ltd. and Sandoz Ltd, both Swiss companies with a long history of pharmaceutical experience. Ciba-Geigy was formed in 1970 by the merger of J. R. Geigy Ltd (founded in Basel in 1758) and Ciba (founded in Basel in 1859). Combining the histories of all mering partners involved, the company's effective history spans over 250 years.
Ciba-Geigy
Johann Rudolf Geigy-Gemuseus (1733–1793) began trading in 1758 in materials, chemicals, dyes and drugs of all kinds"[cite this quote] in Basel, Switzerland. Johann Rudolf Geigy-Merian (1830–1917) and Johann Muller-Pack acquired a site in Basel in 1857, where they built a dyewood mill and a dye extraction plant. Two years later, they began the production of synthetic fuchsine. In 1901, they formed the public limited company Geigy and the name of the company was changed to J. R. Geigy Ltd in 1914.
In 1859 Alexander Clavel (1805 – 1873) took up the production of fuchsine in his factory for silk-dyeing works in Basel. In 1864, a new site for the production of synthetic dyes was constructed, and in 1873, Clavel sold his dye factory to the new company Bindschedler & Busch. In 1884 Bindschedler & Busch is transformed into a joint-stock company with the name "Gesellschaft für Chemische Industrie Basel" (Company for Chemical Industry Basel). The abbreviation Ciba was adopted as the company's name in 1945.
In 1925 J. R. Geigy Ltd. began producing textile auxiliaries,[clarification needed] an activity which Ciba took up in 1928.
In 1939, Geigy chemist Paul Hermann Müller discovered that DDT was effective against malaria-bearing insects. He received the Nobel Prize in Medicine for this work in 1948.
Ciba and Geigy merged in 1971 to form Ciba‑Geigy Ltd., and this company merged with Sandoz in 1996 to form Novartis.
Sandoz
Founded in 1886 by Dr. Alfred Kern (1850–1893) and Edouard Sandoz (1853–1928). The first dyes manufactured there were alizarine blue and auramine. After Kern's death, the partnership became the corporation Chemische Fabrik vormals Sandoz in 1895. The company began producing the fever-reducing drug antipyrin in the same year. Further pharmaceutical research began in 1917 under Professor Arthur Stoll (1887–1971). In 1899, the company began producing the sugar substitute saccharin.
Between the World Wars, Gynergen (1921) and Calcium-Sandoz (1929) were brought to market. Sandoz also produced chemicals for textiles, paper, and leather, beginning in 1929. In 1939, they began producing agricultural chemicals.
In 2005, Sandoz expanded significantly though the acquisition of Hexal, one of Germany's largest generic drug companies, and Eon Labs, a fast-growing US generic pharmaceutical company.
Sandoz opened its first foreign offices in 1964.
In 1967, Sandoz merged with Wander AG (known for Ovomaltine and Isostar). Sandoz acquired the companies Delmark, Wasabröd (a Swedish manufacturer of crisp bread), and Gerber Products Company (a baby food company).
On 1 November 1986, a fire broke out in a production plant storage room, which led to a large amount of pesticide being released into the upper Rhine river. This exposure had quite a negative impact on the aquatic and environmental surroundings.
In 1995, Sandoz spun off its speciality chemicals business to form Clariant. Subsequently, in 1997, Clariant merged with the speciality chemicals business that was spun off from Hoechst AG in Germany.
After the merger
After the merger, Novartis reorganized its activities, and spun out its chemicals activities as Ciba Specialty Chemicals (now a part of BASF).
In 1998 the company made headlines with its biotechnology licensing agreement with the UC Berkeley Department of Plant and Microbial Biology. Critics of the agreement expressed concern over prospects that the agreement would diminish academic objectivity, or lead to the commercialization of genetically modified plants. The agreement expired in 2003.
Novartis combined its agricultural division with that of AstraZeneca to create Syngenta in November 2000.
In 2003, Novartis created a new company named Sandoz, a subsidiary that bundles its generic drug production, reusing the predecessor brand.
On 20 April 2006, Novartis acquired the California-based Chiron Corporation. Chiron was formerly divided into three units: Chiron Vaccines and Chiron Blood Testing, which now combine to form Novartis Vaccines and Diagnostics, and Chiron BioPharmaceuticals, to be integrated into Novartis Pharmaceuticals.
The ongoing Basel Campus Project has the aim to transform the St. Johann site - Novartis headquarters in Basel - "from an industrial complex to a place of innovation, knowledge and encounter".[11]
In 2005, Novartis introduced Certican (Everolimus), an immunosuppressant, and in October 2006 began marketing Telbivudine, a new antiviral drug for hepatitis B."
Historical overview taken from:[3] and [4]
Financial Highlights
(In USD millions, unless indicated otherwise)
2007
Total Group net sales: 39 800
Continuing operations
- Net sales: 38 072
- Operating income excluding environmental and restructuring charges: 7 815
- Return on net sales (%): 20.5
- Operating income: 6 781
- Net income: 6 540
Net income – Discontinued operations 5 428 377
Net income – Total Group 11 968 7 202
Basic earnings per share:
- Continuing operations: 2.81
- Total Group: 5.15
R&D investments: 6 430
- As % of net sales: 16.9
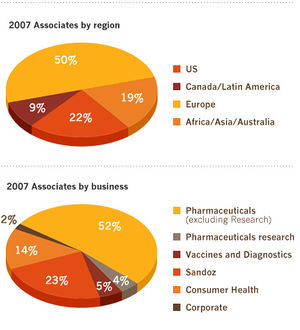
Number of associates: 98 200
2006
Total Group net sales: 37 020
Continuing operations:
- Net sales: 34 393
- Operating income excluding environmental and restructuring charges: 7 642
- Return on net sales(%): 22.2
- Operating income: 7 642
- Net income: 6 825
Net income – Discontinued operations: 377
Net income – Total Group: 7 202
Basic earnings per share:
- Continuing operations1: 2.90
- Total Group: 3.06
R&D investments1: 5 321
- As % of net sales: 15.5
Number of associates: 94 241
For more information please view the 2007 Novartis Annual Financial report, found at: [5]
Business Strategy
Novartis' business strategy can be outlined through its 6 major product-lines:
PHARMACEUTICALS[6]
The company's Pharmaceuticals portfolio includes more than 45 key marketed products, among which the following can be found:
Diovan HCT/Co-Diovan (valsartan and hydrochlorothiazide)
Diovan HCT/Co-Diovan (valsartan and hydrochlorothiazide) For hypertension.
For more information, visit www.diovan.com
Gleevec/Glivec (imatinib mesylate/imatinib)
Gleevec/Glivec (imatinib mesylate/imatinib)
Marketed to treat certain forms of leukemia, gastrointestinal stromal (GIST) tumors and various rare cancers
More info: Visit www.glivec.com
Lamisil (terbinafine)
Lamisil (terbinafine)
Antifungal agent
More info: www.lamisil.com
Zometa (zoledronic acid)
Zometa (zoledronic acid)
Special for cancers that have spread (or "metastasized") to the bones from solid tumors
Pleas view: www.zometa.com or www.us.zometa.com (US residents only)
VACCINES AND DIAGNOSTICS
Novartis' Vaccines and Diagnostics Division has specialized in the development of products to combat over 20 vaccine-preventable viral and bacterial diseases, as well as an array of sophisticated blood-testing equipment.
The current portfolio includes vaccines for:
* Influenza * Meningitis * Rabies * Tick-borne encephalitis * Haemophilus influenzae type B (Hib) * Polio * Diphtheria * Tetanus * Pertussis (whooping cough)
Novartis is the world’s fifth-largest vaccines manufacturer and the second-largest supplier of influenza vaccines in the US.[7]
SANDOZ
Sandoz is the generic pharmaceuticals branch of Novartis, specializing in distributing and manufacturing a vast array of low-cost pharmaceutical products no longer protected by patents.
"The key product groups of Sandoz include antibiotics, treatments for central-nervous-system disorders, gastrointestinal medicines, cardiovascular treatments and hormone therapies."[8]
OVER-THE-COUNTER
"Over-the-Counter (OTC) is a world leader in the research, development, production and marketing of self-medication products that do not require prescriptions."[9]
ANIMAL HEALTH
"Animal Health focuses on the well-being of companion animals and on the health and productivity of farm animals. Our product range provides solutions for the prevention and treatment of several widespread animal diseases and parasite infestations."[10]
CIBA VISION
CIBA VISION specializes in contact lenses and lens-care production in over 70 countries[11]
Corporate Accountability
Labor
"Novartis - dealing with complexities of implementing a living wage"
"Based in Basel, this Swiss pharmaceutical company employs more than 101,000 people and operates in 140 countries. Its focus extends not only to the users of its products but to its employees as well being one of the first international companies to develop and implement a voluntary commitment to pay a living wage to all of its employees around the world. According to Novartis, to focus on a living rather than a minimum wage can contribute to stability and prosperity in communities and attract more skilled, productive and loyal employees. However, being one of the first companies to implement a living wage meant that it was done in the absence of universally accepted models. Novartis took a range of existing methodologies that considered specific geographic data and household expenditure for varying household sizes and places of residence."
Human Rights
"Misdeeds by pharmaceutical industry"
29-11-2006
"New medicines are often tested on people in developing countries in an unethical manner. The Wemos Foundation in Amsterdam, the Netherlands, finds it scandalous that the pharmaceutical industry exploits the weak position of people in developing countries. A new report from the Dutch Centre for Research on Multinational Corporations (SOMO) documents no fewer than 22 known cases in which the rights of trial subjects have been violated.
The report was released on Friday, 17 November during an event held to mark the silver jubilee of the Wemos Foundation. Liesbeth van der Kruit took such politicians as Diederik Samsom (PvdA) and Kees Vendrik(GroenLinks) to task about this issue during the Grote Verkiezingsshow (the Great Election Show). The report describes tests that involved various well-known pharmaceutical companies such as Novo Nordisk, Pfizer, Johnson & Johnson, Novartis, AstraZeneca and GlaxoSmithKline."[12]
"Compulsory licensing “a last mechanism,” says Thai Health Minister"
August, 2008. "Thailand’s new Public Health Minister, Chawarat Charnveerakul, has said that imposing compulsory licensing (CL) is “the last mechanism” that the government would use “to help the poor get access to life-saving drugs at a fair price."
"First, we have to negotiate with drug firms to reduce the price of their drugs. If they do not agree with us we will then impose compulsory licensing for expensive drugs to save our patients’ lives," said Mr Chawarat, who was appointed to his post this month in a cabinet reshuffle. He added that his Ministry had not revoked the CL policy as a way of obtaining essential drugs, but retained it as “a key mechanism” in negotiating for lower prices."[13]
Environment
USA: Novartis Phasing Out Genetically Engineered Foods
Environment News Service
August 4th, 2000
"Novartis, one of the world's leading producers of genetically engineered seeds, has been phasing out genetically engineered ingredients in its food products worldwide for over a year.
Responding to a statement issued by environmental group Greenpeace International yesterday, Novartis Consumer Health U.S. vice president Sheldon Jones told ENS there is nothing new about the company's stand on genetically engineered food.
Greenpeace cited a letter sent by Novartis' European Consumer Health department as evidence the company had stopped producing food containing genetically engineered ingredients in its own brands on June 30.
In particular, the letter stated production of the candy bar Cereal Chocosoja had been stopped because Novartis could not guarantee its non-GM quality."[14]
Consumer Protection and Product Safety
"INDIA: Novartis Patents Case Far From Dead"
Inter Press Service News Agency
August 9th, 2007
"Cancer patients in India have reason to be relieved at a high court ruling this week which dismissed a petition by Swiss pharmaceuticals multinational corporation (MNC) Novartis challenging an Indian law which denies patents for minor or trivial improvements to known drugs. At immediate stake is the cost of a leukaemia drug, imatinib mesylate. Novartis prices its brand of the medicine, Gleevec/Glivec, at Rs 120,000 (3,000 US dollars) per dose. Indian generic drug manufacturers sell it at Rs 8,000 (200 dollars). India’s average per capita annual income is equivalent to only a fifth of the price of a single dose of Gleevec/Glivec. Had Novartis been granted a patent on its version of the drug, tens of thousands of Indians would have been deprived of life-saving treatment.[15]
"The Berlin medical journal "arznei-telegramm" accused Novartis in 2002 of leaving out unfavourable results in the publication of a study of the drug Diovan, in order to make the efficacy of the drug look better than it actually was. The same journal also accused Novartis of illegal marketing practices and creating expectations of efficacy that could not be met."[16]
"In 2002, the Swiss consumer protection agency Stiftung für Konsumentenschutz criticized Novartis for misleading consumers. Novartis had stated in its sales promotion that its drug Mebucasol F was new on the market, but the active ingredients would be the same as those of an older but cheaper drug with the name Sangerol."[17]
Anti-Trust and Tax Practices
Farmers Launch Anti-Trust Suit Against Monsanto
January 4th, 2000
"Six farmers -- from the U.S. and France -- named as representatives of farmers worldwide, under the aegis of the National Family Farm Coalition, in a suit formulated by Cohen Milstein Hausfeld & Toll on behalf of a consortium of other firms, have launched a major anti-trust, price fixing law suit against the Monsanto Corp. and nine corporate co-conspirators (including Novartis)"[18]
In 2004, a class action lawsuit was filed in the US against Novartis accused them of providing fraudulent kickbacks, discounts and rebates to encourage pharmacy benefits managers to put its drugs on their formularies. The case is still pending.[19]
Social Responsibility Initiatives
Governance
Board of Directors
Daniel Vasella, M.D: Chairman and CEO
Ulrich Lehner, Ph.D: Vice Chairman and Lead Director
Hans-Joerg Rudloff: Vice Chairman
Peter Burckhardt, M.D.
Srikant Datar, Ph.D.
William W. George
Alexandre F. Jetzer
Pierre Landolt
Andreas von Planta, Ph.D.
Dr. Ing. Wendelin Wiedeking
Marjorie M. Yang
Rolf M. Zinkernagel, M.D.
Ann Fudge
Executive Comittee
Daniel Vasella, M.D.: Chairman and CEO
Andreas Rummelt, Ph.D.: Group Head of Quality Assurance and Technical Operations
Joseph Jimenez: CEO Pharmaceuticals
Joerg Reinhardt, Ph.D.: COO of the Novartis Group
Juergen Brokatzky-Geiger, Ph.D: Head of Human Resources
Mark C. Fishman, MD: President of the Novartis Institutes for Biomedical Research (NIBR)
Raymund Breu, Ph.D.: Chief Financial Officer
Thomas Wellauer, Ph.D.: Head of Corporate Affairs
Thomas Werlen, Ph.D: General Counsel
Permanent Attendee of the Executive Committee
Andrin Oswald: CEO Vaccines and Diagnostics
David Epstein: President and CEO Oncology and Molecular Diagnostics
George Gunn: CEO Consumer Health, President and CEO Animal Health
Jeffrey George
Contact Information
Head Office
Novartis International AG CH-4002 Basel Switzerland
Tel: +41 61 324 11 11
Fax: +41 61 324 80 01
Monday - Friday, 8:30 - 17:00, GMT+1 (Central European Time)
Articles and Resources
Books on the Company
Related SourceWatch Articles
Sources
- ↑ "About Novartis: Facts"
- ↑ "Novartis completes divestment program with transfer of Gerber baby foods business"
- ↑ "Novartis Company History"
- ↑ "Wikipedia article on Novartis"
- ↑ "2007 Novartis Annual Financial report"
- ↑ "Novartis Pharmaceuticals Division"
- ↑ "Novartis' Vaccines and Diagnostics Division"
- ↑ "About Sandoz: Novartis' generic pharmaceuticals division"
- ↑ "Novartis' Over-the-counter product line"
- ↑ "Novartis Animal Health"
- ↑ "CIBA Vision"
- ↑ "Report from the Dutch Centre for Research on Multinational Corporations on unfair pharmaceutical trial practices"
- ↑ "Compulsory licensing on pharmaceutical drugs in Thailand"
- ↑ "Novartis and Genetically Engineered Foods"
- ↑ "Novartis denied patent in India"
- ↑ "More Consumers International Claims on Novartis"
- ↑ "More Consumers International Claims on Novartis"
- ↑ "Farmers Launch Anti-Trust Suit Against Monsanto and Novartis"
- ↑ "Consumers International's list of claims against Novartis"
External Resources
Consumers International: http://www.consumersinternational.org
Business and Human Rights: http://www.business-humanrights.org
Corporate Watch: http://www.corpwatch.org
Centre for Research on Multinational Corporations: http://somo.nl/